
Aug 3, 2023 | Drug and Device Corner, Drugs, Medical Devices
Thinking about an FDA gap assessment this year? Contact EAS now to schedule your 2023 audit. Schedules toward the end of the year tend to book up suddenly, now is the time to lock in your audit, whether it be onsite or virtual. We would be happy to prepare an audit...

Aug 3, 2023 | Cannabis, FDA and USDA Regulatory Update
On July 27, 2023, the U.S. Congress requested information to guide the assessment of the potential for a regulatory pathway for hemp-derived Cannabidiol (CBD) products with the goal to prioritize consumer safety and bring certainty to the U.S. market. Since the...
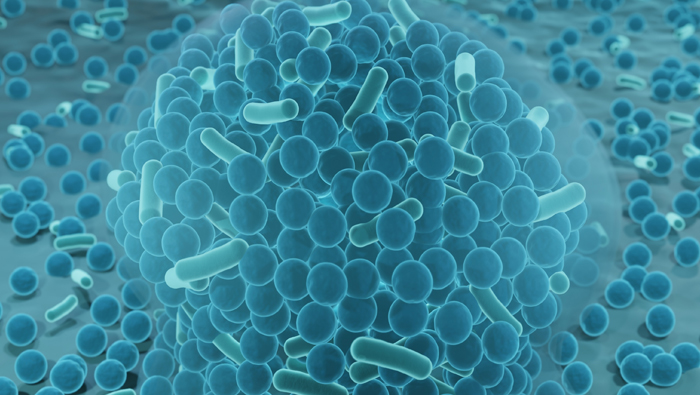
Jul 5, 2023 | Issue of the Month, Medical Devices
Biofilms and Healthcare By David W Koenig, PhD., EAS Consulting Group Independent Consultant Healthcare facilities — such as hospitals, nursing homes and outpatient facilities — are opportunistic locations for acquiring secondary infections unrelated to a...

Jun 27, 2023 | Drug and Device Corner, Drugs, Medical Devices
The FDA has published a new Guidance Document Assessing User Fees Under the Generic Drug User Fee Amendments of 2022 which explains in great detail the GDUFA program user fee obligations. GDUFA III made slight changes to the user fee structure. (API facility fees...

Jun 5, 2023 | Foods, Issue of the Month
By Rafael Olivares, EAS Independent Consultant A career in food safety is not an easy one. It is a stressful profession that requires dedication and a commitment to learn continuously and to teach, to communicate effectively to an audience, in most cases, obstinate,...

May 31, 2023 | Drugs, FDA and USDA Regulatory Update
A recent FDA warning letter concerning identity testing and contaminated glycerin highlights the significance of a recurring potential public health hazard. After several lethal poisonings worldwide, it is apparent that high-risk drug components contaminated with...






